By
 “The U.S. Food and Drug Administration (FDA) on Wednesday amended the Emergency Use Authorizations (EUAs) for the new Pfizer and Moderna COVID-19 Omicron booster shots for children as young as 5 years old — despite having no direct data on the safety or effectiveness of the shots in children.”
“The U.S. Food and Drug Administration (FDA) on Wednesday amended the Emergency Use Authorizations (EUAs) for the new Pfizer and Moderna COVID-19 Omicron booster shots for children as young as 5 years old — despite having no direct data on the safety or effectiveness of the shots in children.”
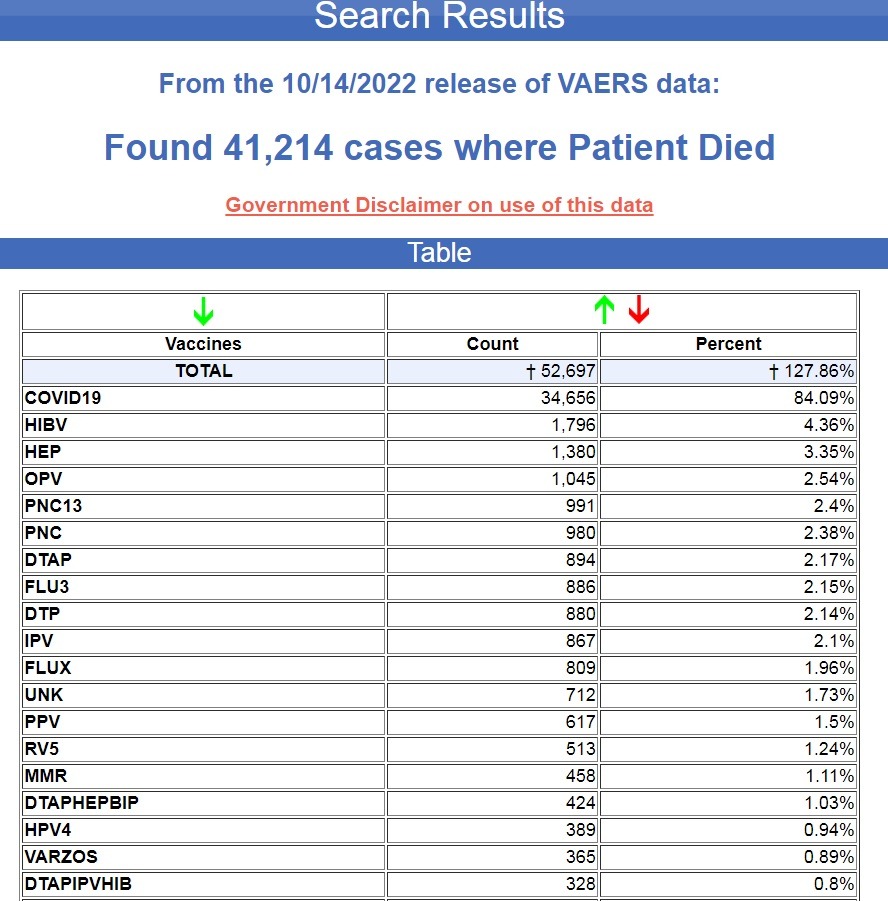 “Since the emergency use authorization of the COVID-19 vaccines in December of 2020, through the latest update of the VAERS database on October 14, 2022, 84% of all deaths reported after vaccination for the past 31+ years have been reported following COVID-19 vaccines. ”
“Since the emergency use authorization of the COVID-19 vaccines in December of 2020, through the latest update of the VAERS database on October 14, 2022, 84% of all deaths reported after vaccination for the past 31+ years have been reported following COVID-19 vaccines. ”